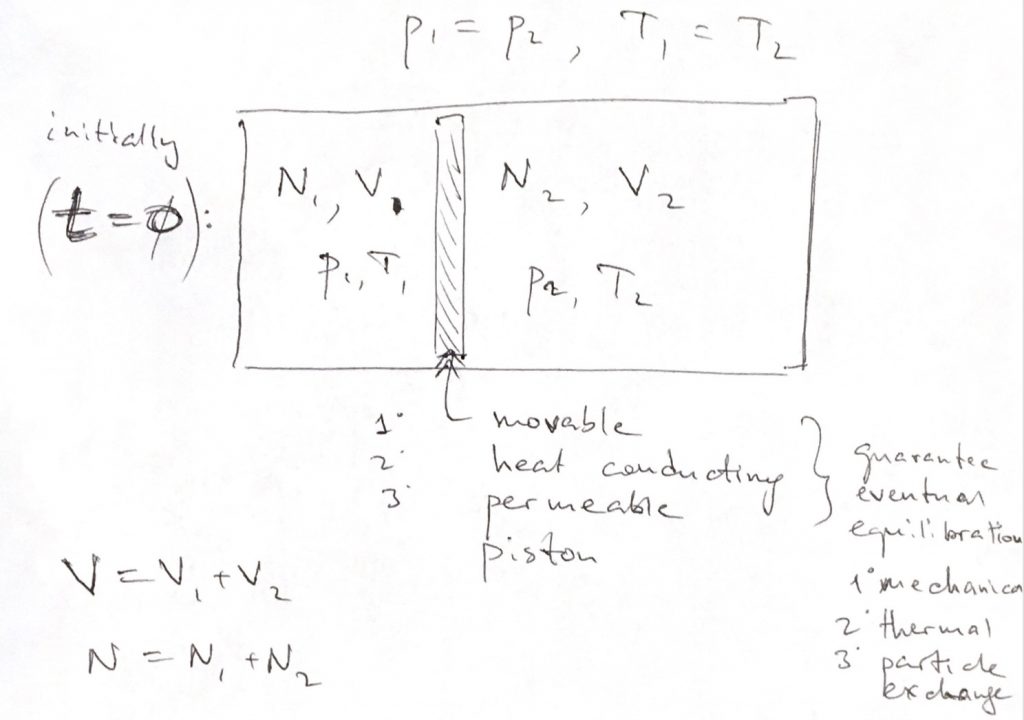
Consider a box partitioned into volumes V1 V 1 and V2 V 2 both of which are kept at the same temperature and pressure. 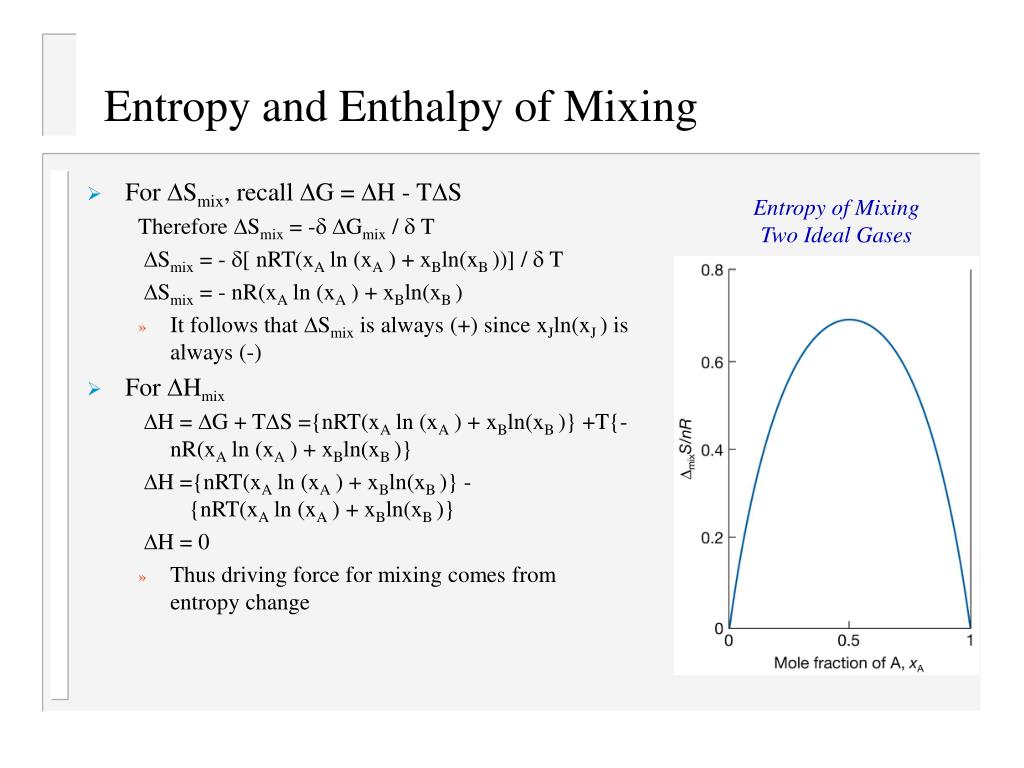
Faber 30 and Kleppa 31 attempted to find volume-based formulae for the excess entropy of. Thermodynamics parameters like enthalpy of mixing ( H mix), entropy of mixing (S mix), and topological parameters like (Zhang et al., 2008b) and mismatch entropy (S ) were used (Takeuchi et al., 2013a) to demarcate the BMG and solid solution formation conditions in multicomponent alloys. However, I have a seeming counterexample at hand. In addition, some efforts have been made to find the excess entropy of mixing of different phases. This concept of entropy as a measure of disorder will become increasingly apparent if you undertake a study of statistical mechanics. For small deviations from the above conditions, the excess entropy of mixing was proportional to the product of the mole fractions of the. In this section we are going to calculate the increase on entropy when two different sorts of molecules become mixed, without any reference to the flow of heat. This simulation was made at the University of Colorado Boulder, Department of Chemical and Biological Engineering. There is a general notion that the entropy of mixing should always be positive (or zero if we are mixing exactly the same stuff). The theoretical entropy of mixing of liquids was found to be ideal only if the diameters of the molecules are the same, the molar volumes of the components are equal, and if the volume of mixing is zero. This simulation runs on desktop using the free Wolfram Player. The entropy of mixing is defined as the increase of disorder in a multi-component system upon transition from a fully demixed (partitioned) to an ideally mixed. When the initial pressures of A and B are equal and the “remove barrier” option is selected, which corresponds to mixing at constant pressure, the entropy of mixing is: \[ \Delta S_ \) are the mole fractions of A and B in the final mixture. As the number of moles increases, the color becomes more intense. Gas A is colored red and gas B is colored blue, and when the gases mix, different shades of purple result, depending on the ratio of moles of each species. The total entropy change is the sum of the entropy changes of each gas. For the “compress right” option, if the partial pressure of a gas does not change, its entropy does not change, even when mixed with another gas. When the partial pressure decreases, entropy increases. Entropy of mixing is entropy due to the presence different types of atoms in the solid solution and is represented as Smix 8.314(XAln(XA)+(1-XA)ln(1-XA)). For the “remove barrier” option, the entropy change of each gas is the same as that of a gas expanding into a vacuum. 
Select the “mix gases” play button to initiate mixing. Mixing is always a spontaneous process for an ideal solution.In this simulation, ideal gases A and B are mixed isothermally by keeping the total volume constant (“remove barrier” option) or by adding gas A to gas B so the final volume is the same as the initial volume of B (“compress right” option). Also, these interactions may make the volume non-additive as well (as in the case of ethanol and water). But for many combinations of liquids or solids, the strong intermolecular forces may make mixing unfavorable (for example in the case of vegetable oil and water). To gain some intuition about entropy, let us explore the mixing of a multicomponent ideal gas. 2.This figure shows integral entropy of formation, f S, from two assessments for Al-Ni alloys at 1,500 K. Of course, such a definition should be supplemented by a more precise definition of disorder after all, one man’s trash is another man’s treasure. Another example, which shows both the integral formation and mixing quantities is given in Fig. Entropies of gases are much larger than those of condensed phases. The entropy also increases as the pressure or concentration becomes smaller. Traditionally, the increment of configuration entropy with temperature was computed by time-consuming thermodynamic integration methods. The entropy of a substance increases with its molecular weight and complexity and with temperature. < 0\) implying that mixing is always a spontaneous process for an ideal solution. Entropy is widely understood as a measure of disorder. entropy of mixing tutorial all along with the key concepts of entropy changes in phase transitions, entropy changes in chemical reactions and standard. Configuration entropy is believed to stabilize disordered solid solution phases in multicomponent systems at elevated temperatures over intermetallic compounds by lowering the Gibbs free energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
